Quantum computing has provided new insights into the fundamentals of photochemical reactions that have proven difficult to study. The results could improve scientists’ understanding of light-driven processes such as photosynthesis, smoke formation and ozone destruction.
Photochemical processes occur when the nuclei of atoms and their electrons take different configurations after absorbing a photon. Some of these reactions are guided by a quantum phenomenon called conical intersection, where the potential energy describing a molecule in its ground state and its excited state intersect. In these cases, the perturbation of quantum mechanics can prevent certain molecular transitions from occurring – a restriction called phase geometry. This narrows the path that can be taken into account and affects the outcome of the reaction. Phase geometry was known around the 1950s, but because of the femtosecond timescales involved, it was never directly observed in a molecular system.
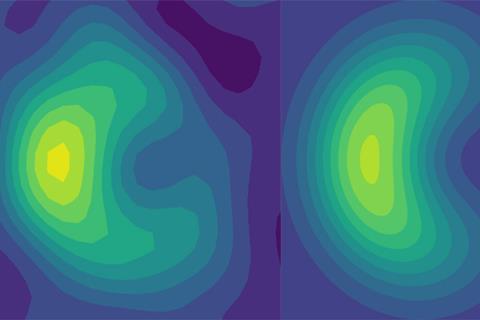
Now, two research groups working independently of each other have shown how phase geometry can be measured using quantum simulators.
‘This phase is very difficult to see real molecules because they occur far from the surface and require a pure quantum state with minimal thermal disturbances,’ says researcher of quantum systems Kenneth Brown from Duke University in North Carolina, US.
‘We are building a quantum system that has some properties of the system we want to study,’ says Brown. The design of this quantum simulator allowed the Duke team to measure the effect ‘in a very simple time to study’, says Brown.
The researchers used a laser to create a chain of five trapped ytterbium ions in a way that mimics the behavior of atoms in a conical compound. As the quantum dynamics of trapped ions are much slower than those of molecules, the team was able to directly measure how the phase geometry affects the spatial distribution of ion currents.
‘Our experiment is one of the first demonstrations of how we can create electron vibrational coupling of trapped ions,’ says Brown. He notes that understanding phase geometry can give chemists ‘another way to control’ the products produced during multi-product reactions.
The Duke team’s findings were published alongside similar work led by researchers at the University of Sydney, Australia. The team, led by Ivan Kassal, used an analog quantum simulator based on a single ion of ytterbium ion.
‘One of the most important things here is that we were able to observe, in real time, the perturbation of the geometric phase of this rapidly behaving system [a] molecular process,’ says Vanessa Agudelo, a PhD student in Kassal’s lab who worked on the project.
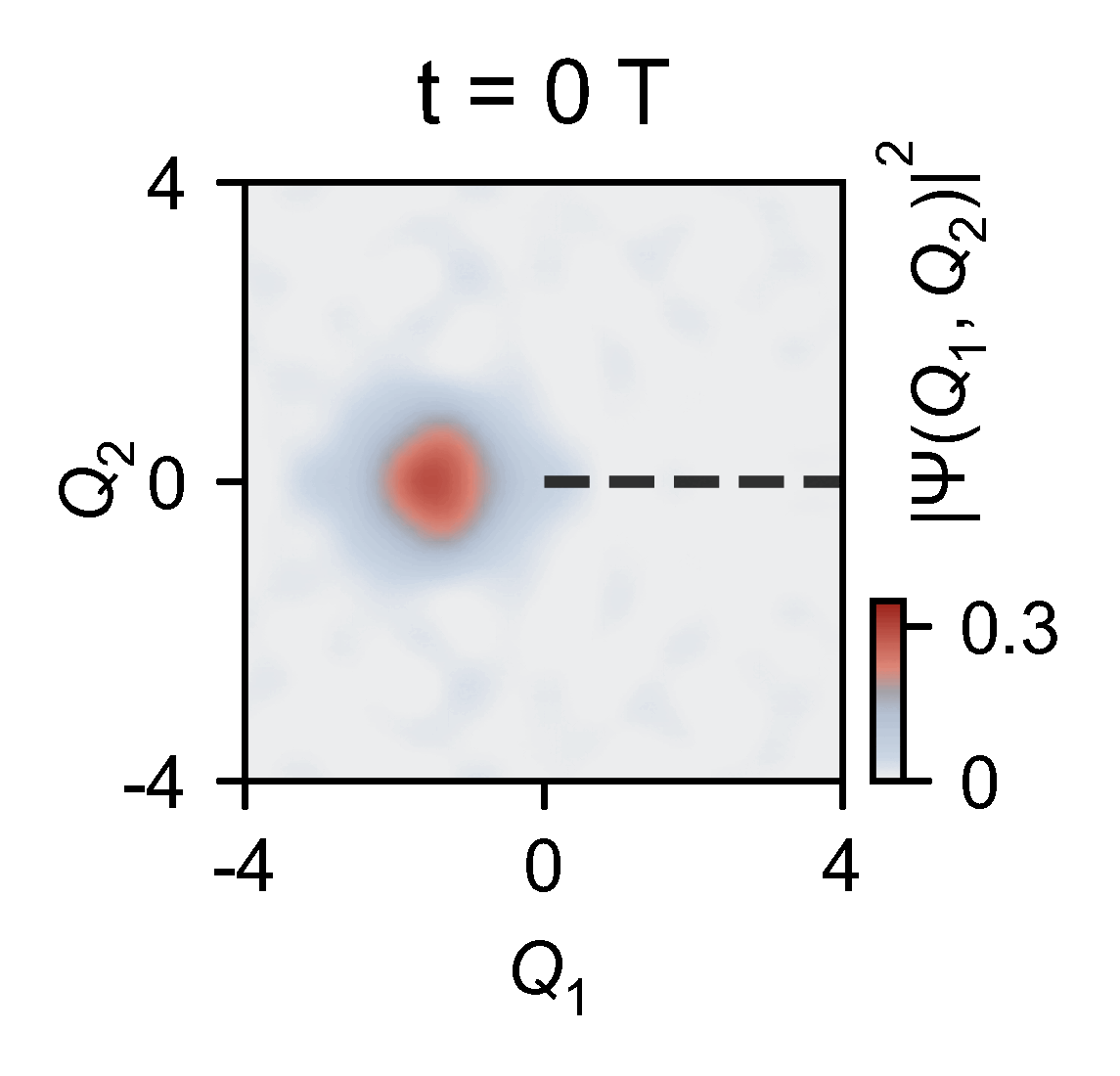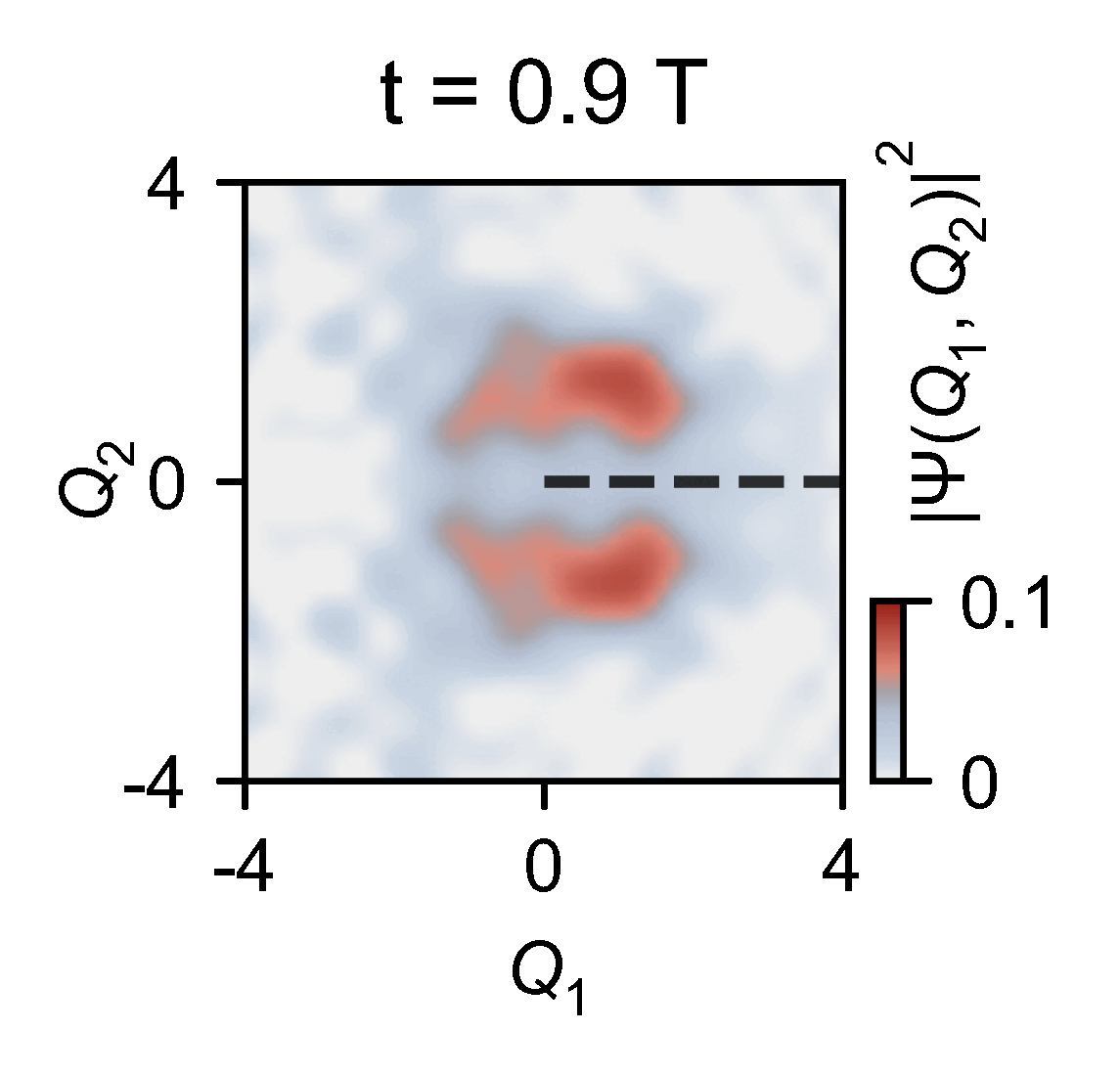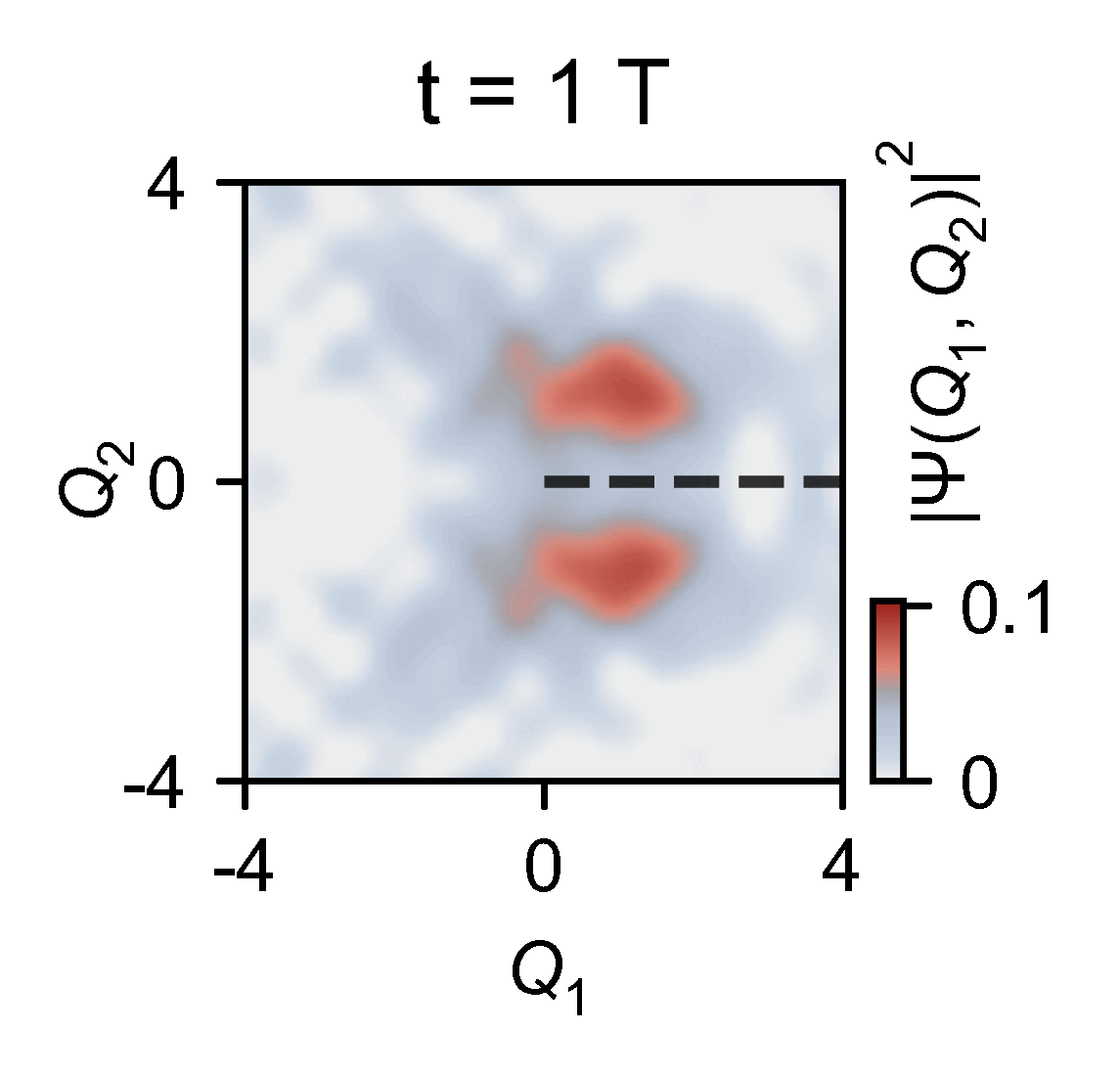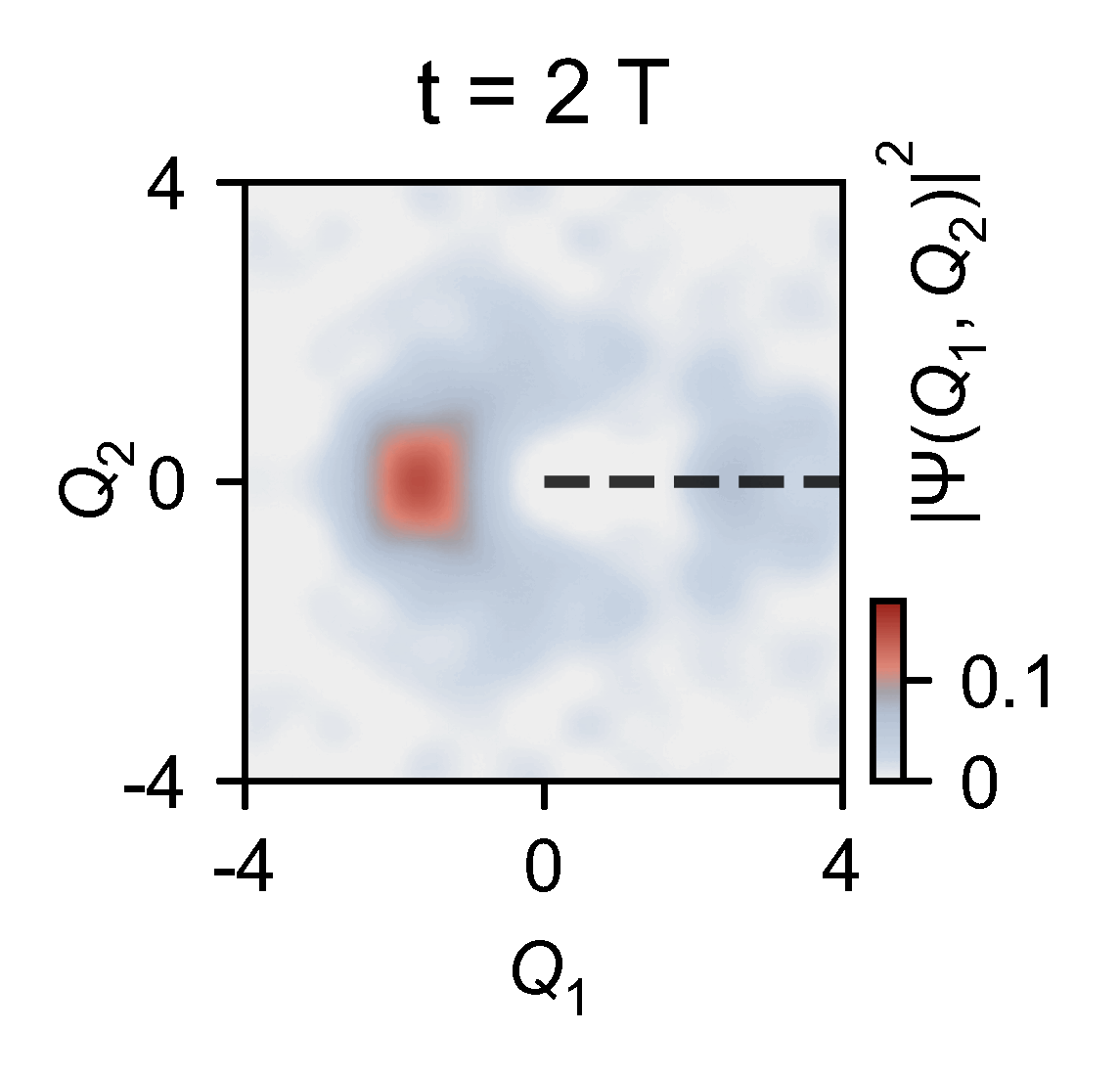
A quantum computer allowed them to slow down the chemical dynamics of the system they were studying from femtoseconds to milliseconds, allowing meaningful observations.
‘Here you have a real video of a single atom split in half, … interfering with itself when it goes to the other side of the conical junction,’ explains Kassal. ‘This measures the incoming photon and how the molecule interacts on femtosecond timescales.’
‘That’s especially important for things like atmospheric chemistry – why does smog appear? How is the ozone layer formed? Or how to be destroyed?’ He added.
While the technical approach taken by the two groups is different, their results are consistent. Kassal notes that the work highlights how quantum computing can help solve chemistry problems. ‘The goal of using quantum computers in chemistry is to be able to simulate any kind of chemical process … like drug discovery, or the discovery of better materials,’ he says.
#Quantum #computing #insights #photochemical #processes